|
Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA. Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA Funding Writing support was provided by Sheri Arndt, PharmD, of C4 MedSolutions, LLC (Yardley, PA, USA), a CHC Group company, funded by Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA. These results support first-line pembro + chemo in pts with metastatic squamous NSCLC. Pembro + chemo continued to demonstrate improved outcomes, including OS, PFS, ORR and PFS2, compared with placebo + chemo for previously untreated metastatic squamous NSCLC, with a manageable safety profile. PFS2 was longer for 1L pembro + chemo (median, 13.8 mo vs 9.1 mo), with no difference by PD-L1 TPS.Table: 2L+ therapy was received by 32.0% in the pembro + chemo arm and 59.4% (49.1% 2L+ antiPD1) in the placebo + chemo arm. With median follow-up of 14.3 mo (range, 0.1–31.3) and 365 total deaths, pembro + chemo continued to improve OS (median, 17.1 vs 11.6 mo), PFS (median, 8.0 vs 5.1 Table) and ORR (62.6% vs 38.4%). PFS2 was an exploratory endpoint and defined as time from randomization to time of PD per investigator or death after start of 2L therapy, whichever occurred first. Response was assessed by RECIST v1.1 by blinded central review. After positive results from IA2, pts on the placebo arm were unblinded to stop placebo. Pts in the placebo + chemo arm could crossover to pembro monotherapy at the time of confirmed PD. Randomization was stratified by PD-L1 TPS (≥1%/<1%), choice of taxane (paclitaxel/nab-paclitaxel), and region (East Asia/rest of world). MethodsĮligible pts were randomized 1:1 to pembro (n = 278) or placebo (n = 281) + 4 cycles of carboplatin and paclitaxel/nab-paclitaxel. We report updated OS from the protocol-specified final analysis and, for the first time, PFS2. Benefit was observed irrespective of PD-L1 TPS. In the randomized KEYNOTE-407 study (NCT02775435), pembro + carboplatin and paclitaxel/nab-paclitaxel (chemo) significantly improved OS, PFS, and ORR compared with placebo + chemo in patients (pts) with previously untreated metastatic squamous NSCLC. 18 Oncology, Montefiore Medical Center/Albert Einstein College of Medicine, Bronx/US.16 Department Of Oncology, Cancer Hospital of Jilin Province, Changchun/CN.15 State Healthcare Institute, Pyatigorsk Oncology Dispensary, Pyatigorsk/RU.14 Oncology, Poznan University of Medical Sciences, Poznan/PL.13 Hematology-oncology, Inje University College of Medicine, Busan/KR.12 Respiratory Medicine, Kyushu University Hospital, Fukuoka/JP.11 Oncology Center, Medica Sur Hospital, Mexico City/MX.10 Oncology, Csongrád County Hospital of Chest Diseases, Deszk/HU.9 Department Of Pulmonology, Veszprém Megyei Tüdőgyógyintézet Farkasgyepű, Farkasgyepu/HU.8 Department Of Medical Oncology, Trakya University, Edirne/TR.7 Medizinische Universitätsklinik Innere Medizin Ii, Universitätskinikum Tübingen, Tübingen/DE. 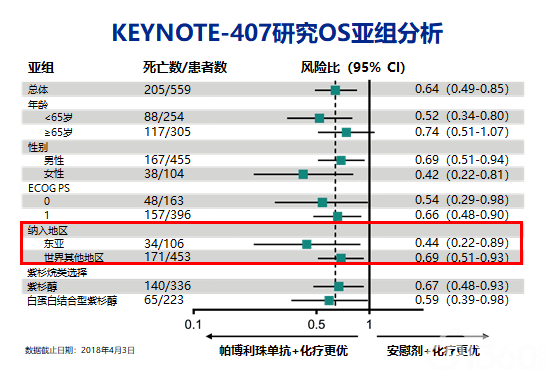
6 Thoracic Oncology Unit, Respiratory Disease Department, Hôpital Larrey, Centre Hospitalier Universitaire Toulouse, Toulouse/FR.5 Medical Oncology, AOU Policlinico Vittorio Emanuele, Catania/IT.4 Cancer Centre Of Southeastern Ontario, Kingston General Hospital, Kingston/CA. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
